Living Materials from Kombucha & CRISPR for a Fungus - 2021.01.19
Plus: Expanding the alphabet of proteins.
☀️ Good morning
Yesterday was Martin Luther King Jr. Day. Hope you enjoy this Tuesday newsletter.
It’s Alive! Materials from Kombucha
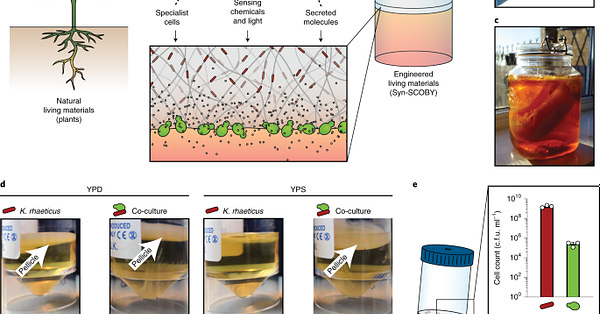
If you ever make kombucha at home (there are ample starter kit options online), you will slowly begin to see a thick layer of goop form on top of your tea. That goop is a pellicle of cellulose, created by acetic acid bacteria in your fermentation mixture. Yeast, mixed in with the tea, make the kombucha “bubbly” and produce carbon dioxide and ethanol. Together, the yeast and bacteria help to create the distinctive flavor profile of kombucha. But they can also be used for so much more.
For a new study in Nature Materials, the Tom Ellis and Tim Lu laboratories at Imperial College and MIT, respectively, teamed up to create “living materials” inspired by kombucha. They used two organisms amenable to genetic engineering to create a synthetic “kombucha” co-culture: a lab strain of budding yeast (Saccharomyces cerevisiae) and a bacterial strain that can produce cellulose, called Komagataeibacter rhaeticus, isolated from kombucha tea.
The bacterial cells make a thick, cellulose pellicle in just three or four days at room temperature. The researchers devised some clever tricks to get the yeast to “float”, suspended, in the cellulose pellicles made by those bacteria. Then, the researchers could engineer the yeast cells to sense environmental contaminants or light, for example, and output signals in response. In this way, they’ve created a synthetic community of cells that work together to create living, programmable materials. That’s pretty cool.
If you’d like to learn more about this study, I recommend the “behind-the-paper” perspective written by one of the authors.
Unleashing the Power of Fungi
Fungi are a veritable smorgasbord of bioactive compounds. Cephalosporin comes from a fungus, as does the first antibiotic to be discovered, penicillin. Unfortunately, it is really difficult to work with some fungi in the laboratory, as many of their genes are only “switched” on while they grow in the wild. In the Amazon rainforest, for instance, you might discover a fungus that produces some great new medicine to cure warts. But take that fungus back to the lab, and it may stop making that medicine entirely—the fungus shuts down.
For a new study, published in Scientific Reports (Open Access), researchers built a CRISPR-based gene activation tool exclusively for fungi. The gene activation technology uses a “deactivated” form of Cas9 that can recognize, but not cut, DNA, fused to an activator protein called VPR. The resulting “fusion” protein, called dCas9-VPR, was then inserted into fungal cells using a method “compatible with several filamentous fungal species.”
As a proof of concept, the researchers used the CRISPR activation system to “switch on” genes that produce a chemical called macrophorin, in a type of fungus called Penicillium rubens. Cool back story—that’s the same fungus that inadvertently helped Alexander Fleming discover penicillin!
A Marvel of Protein-Making Machines
Ribosomes are molecular monstrosities; consisting of dozens of individual proteins, they read through about 60 mRNA “letters” every second, tacking on amino acids to a growing protein at a blistering pace. Most ribosomes use a limited alphabet of just 20 amino acids to build proteins, but bioengineers have been expanding that alphabet in recent decades, bit by bit.
For a new study in ACS Synthetic Biology, researchers created ribosomes in vitro (that is, not inside of cells) that can manufacture proteins that have non-canonical amino acids incorporated at any given position. To make this work, the scientists had to recode the E. coli genome, delete a specific gene called release factor 1, and then break those cells open and harvest their gooey insides. In the end, they were able to use the bacterial goop to build a GFP protein that had two different non-canonical amino acids incorporated at the same time, called p-acetyl-phenylalanine (pAcF) and p-azido-phenylalanine. They extended the work by adding pAcF at one, two, or even five different sites in the protein at the same time. This paper is a nice step forward for cell-free manufacturing of proteins with an expanded alphabet.

🧫 Rapid-Fire Highlights
More research & reviews worth your time
Zip, Zap, Zoop (01100100 01101110 01100001). Researchers at Columbia University took bacterial cells, engineered them with a modified CRISPR system that can sense and respond to redox reactions, and then pulsed them with electricity, pushing the CRISPR system to encode data (in 3-bit units) into CRISPR arrays in the bacterial genomes. Using this method, they could store up to 72 bits of information. It’s direct digital-to-DNA information storage! Nature Chemical Biology. Link
A new paper lays out detailed steps to design and perform base editing experiments in the laboratory, with a focus on mammalian cells. Nature Protocols. Link
A protein called amyloid beta (a short peptide, only about 40 amino acids in length) is the main element of amyloid plaques found in the brains of people with Alzheimer's disease. For a new study, researchers evolved a bacterial enzyme called transpeptidase sortase A that can recognize, and “tag”, a specific amino acid sequence within the amyloid beta proteins. The evolved enzyme was used to detect the amyloid beta protein in very small amounts within human cerebrospinal fluid. Nature Chemical Biology (Open Access). Link
While there are some effective treatments for the herpes simplex virus (HSV-1), they don’t completely eliminate the virus from all of its nooks and crannies. For a new study, researchers used CRISPR-Cas9 to block HSV-1 from replicating—and to eliminate the virus from one of its reservoirs in the eyes—in mice. Nature Biotechnology. Link
This one is amazing—8269 unique, inducible promoters were created in the lab. Each of these sequences had RNA polymerase and LacI repressor binding sites at varying positions, and consisting of varying strengths. Researchers then studied each of the promoter sequences in the lab to better understand how various factors influence the way that a promoter behaves. In the end, they unraveled “practical insights for engineering inducible promoters with desirable characteristics.” Nature Communications (Open Access). Link
The Cas9 protein from Streptococcus pyogenes is a boon to molecular biologists looking to cut and edit a genome, but it’s sorely limited in a major way: it can only make cuts where the letters ‘NGG’ (where N is any of the four nucleotides) already happen to exist in DNA. For a new study, researchers evolved SpCas9, in the laboratory, to instead have a preference for an ‘NAG’ sequence. Nature Communications (Open Access). Link
As “living medicines”—bacteria engineered to go into the gut to diagnose or treat a disease—grow in sophistication, so too must the tools that are used to create them. A new review explains how to edit the genomes of probiotic bacteria, including lactobacilli and bifidobacteria. Current Opinion in Biotechnology (Open Access). Link
Synthetic biology appears to be a scientific priority for the incoming Biden/Harris administration in the United States. In the UK, biological security has long been a topic of conversation. A new article presents 80 questions to consider for UK biological security. PLOS ONE (Open Access). Link
DNA data storage is a hot ticket item right now. A new review lays out the state of the field. Trends in Biotechnology. Link
Cyanobacteria may well be the future of “green” efforts in synthetic biology; they are photosynthetic, after all, and can be engineered to produce fuels or medicines from sunlight and carbon dioxide. A new review takes a look at the state, and current challenges, of cyanobacterial engineering. Trends in Biotechnology. Link
Metabolism is complicated; to learn about it, why not rip some cells open and study their goop-y insides? A new review takes a look at renewable, cell-free biomanufacturing. In other words, how to use cell extracts to produce medicines, or fuels, or nearly anything else you want to make. Current Opinion in Biotechnology. Link
Using E. coli, a new study implemented a “lateral inhibition” gene circuit in which cells near one another inhibit their neighbors. The authors show that the engineered colony is “capable of symmetry-breaking leading to regular spatial patterns during colony growth.” ACS Synthetic Biology. Link
Cells are like snowflakes. Even two genetically-identical cells differ from each other, thanks to random noise in how their proteins are produced, or which genes get turned on at certain times. For a new study, researchers at UCSF created a gene circuit in mammalian cells that can decouple a gene’s mean expression from its noise. This will be an important tool to study how variations between cells—their heterogeneity—impacts their behaviors or traits. Nature Communications (Open Access). Link
Bioengineers often “program” cells with logic gates, which perform operations on some inputs and then return a single output. There are NOR, AND, OR, NAND gates, for example. Cells, though, are not computers; the physical environment in which they live can change the way that these logic gates perform. For a new study, researchers built 20 different NOT gates in living cells, and then grew them in 7 different growth conditions, each of which changed some parameters in how the logic gates operated. Nature Communications (Open Access). Link
To engineer a protein to perform a function not found in nature, scientists introduce mutations, in specific areas, and hope to gradually create proteins that perform the desired task. Mutation Maker is an open-source software to design oligonucleotide sequences to perform mutagenesis for “large-scale protein engineering experiments.” ACS Synthetic Biology (Open Access). Link
For a new study, researchers increased the amount of thiamin (also called vitamin B₁) produced in rice endosperm by engineering just three genes. This is certainly not the first study to create rice that overproduce some essential vitamin or other nutrient; there has been a long push on that front (see the Golden Rice effort for rice engineered to produce vitamin A). Plant Biotechnology Journal. Link
MAGE is an experimental method to engineer the genome en masse. It can be used in both prokaryotic and eukaryotic cells (like E. coli and S. cerevisiae). A new primer explains the method and how it can be applied to diverse scientific problems. Nature Reviews (Open Access). Link
Have a great week.
Until Friday,
— Niko
Bonus Tweet: The study about living materials made from kombucha cultures (featured at top of this newsletter) was a joy to read. For some great photos and a behind-the-scenes look, check out this Twitter thread. 👇

Thanks for reading Cell Crunch, part of Bioeconomy.XYZ. If you enjoy this newsletter, please share it with a friend or colleague.
A version of these newsletters is also posted on Medium. Reach me with tips and feedback @NikoMcCarty or via email.


